United States law stipulates that prescription drugs be tested on animals. The belief is that the injection of an experimental drug into an animal will yield information about that drug’s efficacy, toxicity and safety. It does, but the information is only relevant to the animal species tested in the experiment. This information can not be safely or accurately applied to humans, or any other species for that matter.
Accord to U.S. Secretary of Health and Human Services Michael Leavitt, “Currently, nine out of ten experimental drugs fail in clinical studies because we cannot accurately predict how they will behave in people based on laboratory and animal studies.”1 This was echoed by Food and Drug Administration Commissioner Andrew C. von Eschenbach, MD... "Consider just one stark statistic: Today, nine out of 10 compounds developed in the lab fail in human studies. They fail, in large part because they behave differently in people than they did in animal or laboratory tests.”2
Yet animal-based drug data continue to be generalized to humans. Some researchers will also utilize non-animal tests to determine a drug’s safety and effectiveness, but our government still requires the outdated and unreliable use of animal models. This mandate has cost untold suffering for the animals in laboratories, patients harmed or killed from these drugs, and their loved ones.
THE PHARMACEUTICAL INDUSTRY
The pharmaceutical industry is a business, and like any business its top priority is profit. This misguided motivation leads to irresponsible protocol and eventually to human injury and death.
Click here to learn more about this business.
ANIMAL VICTIMS OF DRUG TESTING
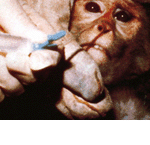
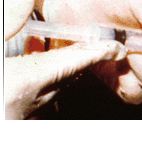
Many animals suffer slow, agonizing deaths in drug testing laboratories—cats, dogs, hamsters, mice, and numerous others. They spend their short lives locked in small, barren cages, given test drugs, observed, killed and dissected. Many are denied food and water. If obvious distress or painful side effects are noticed, they are logged and nothing more; to administer pain relief would be interfering with the study.
Click here to read more.
THE INVALIDITY OF ANIMAL DATA IN DRUG TESTING
The pharmaceutical companies and our government are indeed aware of the potential for inaccurate results when experimenting with animals. This is one of the reasons researchers don’t always rely on them. The other reason is a reluctance to shelve a drug that may market well. So if a drug causes adverse reactions in the animal experiments, it is not automatically tossed aside.
To learn more about how animal data fails as a predictor of human safety and efficacy, click here.
DANGERS OF GENERALIZING ANIMAL DATA TO HUMANS
Adverse drug reactions (ADR) to medications deemed safe for humans based on animal tests seriously injure more than two million people each year. ADRs kill more than 106,000 Americans every year, more than all illegal drugs combined, and are the fourth leading cause of death in America, behind heart disease, cancer and stroke. Perhaps this is because the drug companies are a bit too eager to get their expensive products on the market.
Click here to read examples of how animal testing has caused suffering and death to humans too.
REPLACING ANIMALS IN DRUG TESTING
By utilizing animal replacement methods, which are typically progressive and pass rigorous screening processes, we gain accurate insight into drug safety and value. Abandoning animal experiments will provide valid information, thus saving animals and humans from harm and death.
To learn how research can better predict a drug's effects on humans, click here.
1 Food and Drug Administration press release, FDA Issues Advice to Make Earliest Stages of Clinical Drug Development More Efficient, January 12, 2006.
2Prepared statement for FDA Teleconference: Steps to Advance the Earliest Phases of Clinical Research in the Development of Innovative Medical Treatments, January 12, 2006.
This website is dedicated to the memory of Matt Fancera.